Towards Absolute Zero
Saturday 28th Jul 2012, 4.30pm
Ossie wouldn’t really have survived the ride in our animation, but much of the science is accurate. Here is more background about what’s going on.
Fridge and freezer
Fridges and freezers preserve food because they slow down bacterial growth. In a freezer at -5 C, the rate of growth is negligible.
Water freezes at 0 C
Different substances freeze at different temperatures. For example alcohol (ethanol) freezes at -114.6 C, while nitrogen freezes at -210 C.
Freezing temperature
Temperature is a measure of how much molecules can move – how much kinetic energy they have. In liquids, molecules are packed closely with just enough kinetic energy to escape the attractive forces. As temperature decreases, the energy becomes too low for molecules to move, so freezing may occur. Therefore, freezing temperature partly depends on the strength of attractive forces between molecules.
Helium consists of very light atoms and the attractive forces are small. The atoms never bind strongly enough to overcome the kinetic energy, no matter how low the temperature. Helium can only be made solid by squeezing under 20 times atmospheric pressure at extremely low temperatures.
Temperature scales
In Europe we use the Celsius/Centigrade scale, based on the freezing and boiling points of water (0 C and 100 C). In America, the Fahrenheit scale is more common. Here, 32 F and 212 F are the normal freezing and boiling points of water. For scientists the Kelvin scale is the most useful – you can convert from Celsius to Kelvin by adding 273.15.
Absolute zero
In the Kelvin scale, 0 K (-273.15 C) is absolute zero. At absolute zero, every particle has the minimum amount of energy possible and matter is perfectly ordered. It’s not actually possible to reach this temperature, but it acts as a reference point.
Although particles have minimum energy, it doesn’t mean everything is stationary. Even in a solid at absolute zero, atoms would be moving. For helium, this ‘zero-point energy’ is already large enough to overcome the weak attractive forces, so atoms don’t stick together to form a solid.
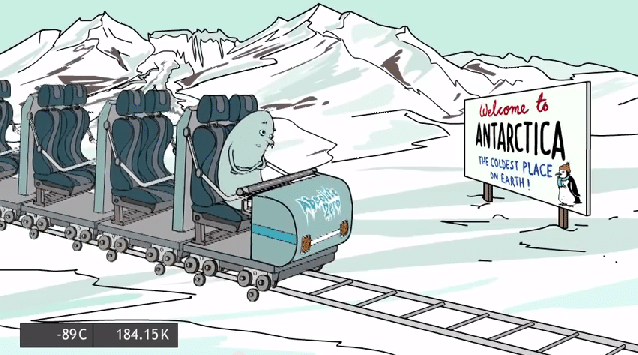
Antarctica
The lowest naturally occurring temperature recorded on Earth was −89.2 C in Antarctica.
Earth’s temperature is a balance between the sun heating the surface and the emission of infrared radiation into space. The planet is tilted on its axis, so in the Antarctic winter the region is pointing away from the sun and is almost entirely dark for about 6 months.
Antarctica is colder than the Arctic. It has a thicker layer of ice protecting it from the warmer ocean underneath, and it sits about 2800 m above sea level.]
Heat flow
Heat is a form of energy. When something cold contacts something hot, the heat energy flows from the hot to the cold.
Molecules in hot objects move more than those in cold objects. When the two objects touch, the energy tends to transfer. The slower molecules of the cold object speed up and the faster molecules of the hot object slow down. Both objects eventually reach the same temperature – ‘thermal equilibrium’. However when the one body is very small (Ossie) and the other is very large (Antarctica), the equilibrium temperature will be almost equal to that of the large body.
Interstellar space
It is commonly assumed that space is a vacuum. However, there are some regions between stars where low density clouds of gas form, which may eventually collapse to form new stars.
In Earth’s atmosphere, 1 cm3 of gas contains about 10^19 (ten billion billion) molecules. In an interstellar gas cloud there are only 100 to 10000 molecules in 1 cm3.
At atmospheric pressure each molecule undergoes about 10^10 (ten billion) collisions per second. In an interstellar gas cloud each molecule collides around once every million seconds (once a fortnight). However, the timescales of space are measured in millions of years, so there is plenty of time for collision.
In space the temperature is only around 20 – 30 K (minus 253 – 243 Celsius). Chemical reactions usually need an energy input to break a chemical bond before another can be made. This is the ‘activation energy barrier’. As the temperature is lowered, molecules have less kinetic energy and the collisions are softer, so reactions may not follow.
In these interstellar regions, many of the molecules are ionized or dissociated by radiation. Simple molecules may exist as positively charged ions or as highly reactive free-radical species. These behave differently from other molecules – in effect a bond has already been broken, so there is no activation barrier. Reactions involving free radicals and ions may actually speed up as the temperature is lowered.
Liquid Helium – zero-viscosity weird
Helium atoms are very light and the forces between them are weak. It only becomes liquid at around 4 K. At around 2K, helium shows a remarkable property – superfluidity.
Normal liquids are characterised by viscosity – how much resistance a fluid has to pushing a solid object through it. Treacle is very viscous, whereas water is less so. In a superfluid, the liquid has zero viscosity.
Superfluid helium in an open container will form a film that can creep up the walls of the container and flow out. With certain types of glass it can also flow through the bottom of a container that would hold normal liquids.
Stark decelerator
Recently, scientists have been devising methods to cool molecules below 1 K. One method is the Stark Decelerator, developed at the Fritz Haber Institute in Berlin, and adapted at Oxford.
In this device a beam of molecules is fired through 100 or more pairs of electrodes. The electrodes exert a repelling force on the molecules that slows them down, losing kinetic energy. By passing enough stages the molecules can be brought almost to a standstill. Temperatures as low as 10 milliKelvin (0.01 K) have been achieved in this way.
Wave-particle duality of light and matter
Einstein, Planck and other early 20th century scientists recognised that although light had previously been thought of as an electromagnetic wave, it could also act as if made up of small packets of energy known as photons.
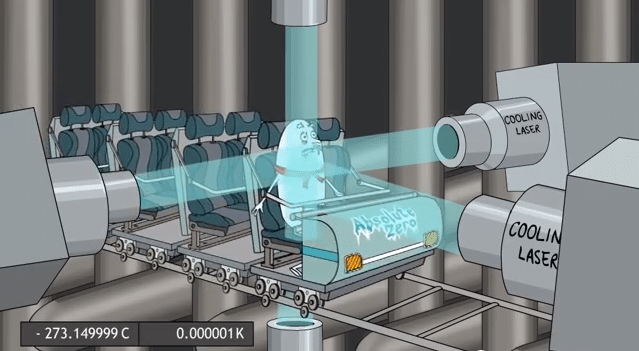
Laser cooling
Normally, when light is absorbed by a gas, individual photons bump into individual atoms. The photon’s energy is transferred to the atom, which is then in an ‘excited state’. The photon is destroyed in this process, but its momentum is taken up by the atom.
In laser cooling, the photon is only absorbed when moving towards the atom, so the atom slows down slightly. By repeating the process many times, the atoms can be brought to a virtual standstill.
Laser cooling experiments use 6 laser beams in perpendicular directions to achieve a slowing of the atoms irrespective of which direction they are moving in.
Implications for chemistry
At low temperatures, wave-particle duality starts to become important. Things that we think of as particles start to behave with wave-like properties. The standard image of molecules bumping into one another is no longer true and we need to consider the properties of waves. This is the field of quantum mechanics, or wave mechanics. Chemical processes are taking place in a completely different environment, so the unexpected may occur. Reactions which wouldn’t be expected due to the high activation energy required may happen because long wavelength waves can penetrate through the activation barrier.
Bose Einstein condensate
At the lowest temperatures, even in low density gases, the wavelength describing the motion of the particles may become so long that it is greater than the average spacing of the particles.
In this situation, the particles may move into a state of matter known as a ‘Bose-Einstein condensate’. Instead of each particle being described by its own wave, the entire sample of particles is described by a single wave.
Further information:
Teaching Resources:
KS5: Beyond the Stars (contact us)
Find out more:
Excerpt from Horizon: What is One Degree?